Search O-GlcNAc sites of proteins, quickly and reliably
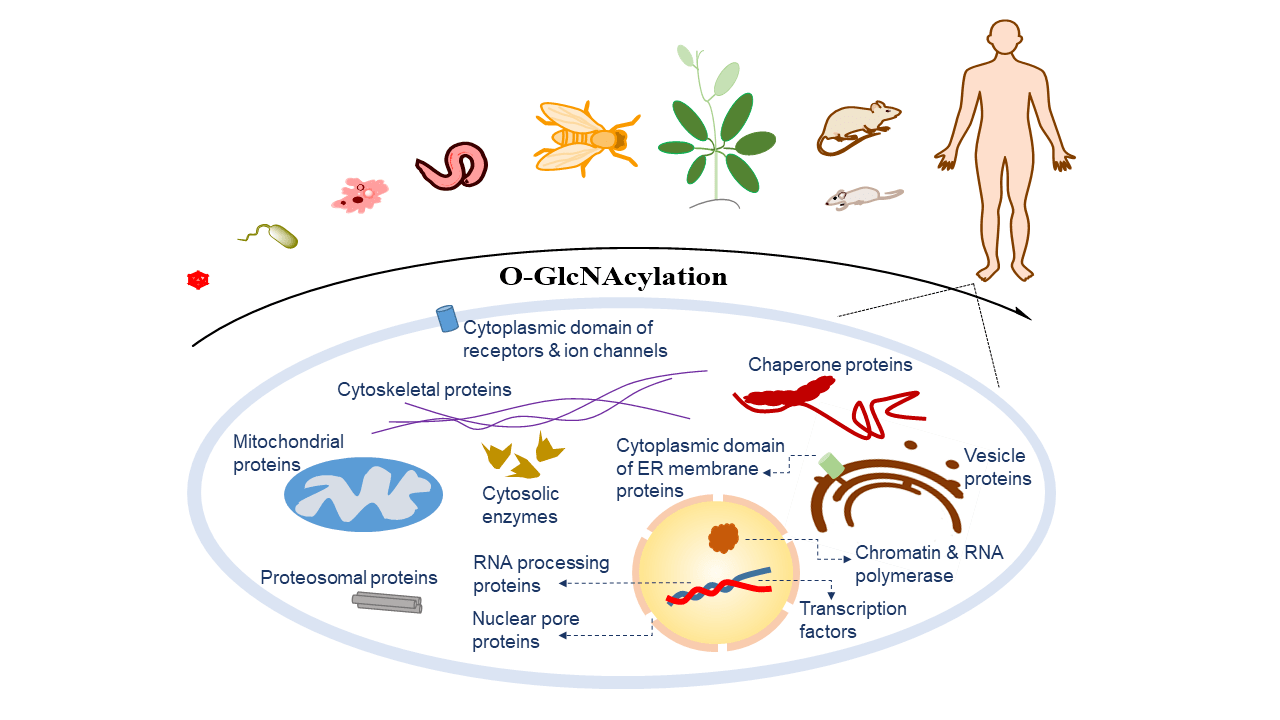
Protein O-GlcNAcylation has been documented in all metazoans (including animals, insects, and plants), some bacteria, fungi, and viruses. O-GlcNAcylation occurs on many proteins in nuclear, cytosolic, and mitochondria. As a critical modification, O-GlcNAc functions in a species-, tissue-/cell-, protein-, and site-specific manner.
If you find this resource helpful, please cite our papers:
Ma J, et al., O-GlcNAcAtlas: A database of experimentally identified O-GlcNAc sites and proteins. Glycobiology. 2021, 31, 7, 719-723, DOI: 10.1093/glycob/cwab003.
Hou C. et al., O-GlcNAcAtlas 4.0: An Updated Protein O-GlcNAcylation Database with Site-specific Quantification. Journal of Molecular Biology, 2025:169033. DOI: 10.1016/j.jmb.2025.169033
The current version: O-GlcNAcAtlas 5.0