oglcnac.org
A curated research portal for O-GlcNAc sites, OGT interaction proteins, glycoform discrimination, and deep-learning site prediction.

Resource Suite
Tools built for discovery workflows
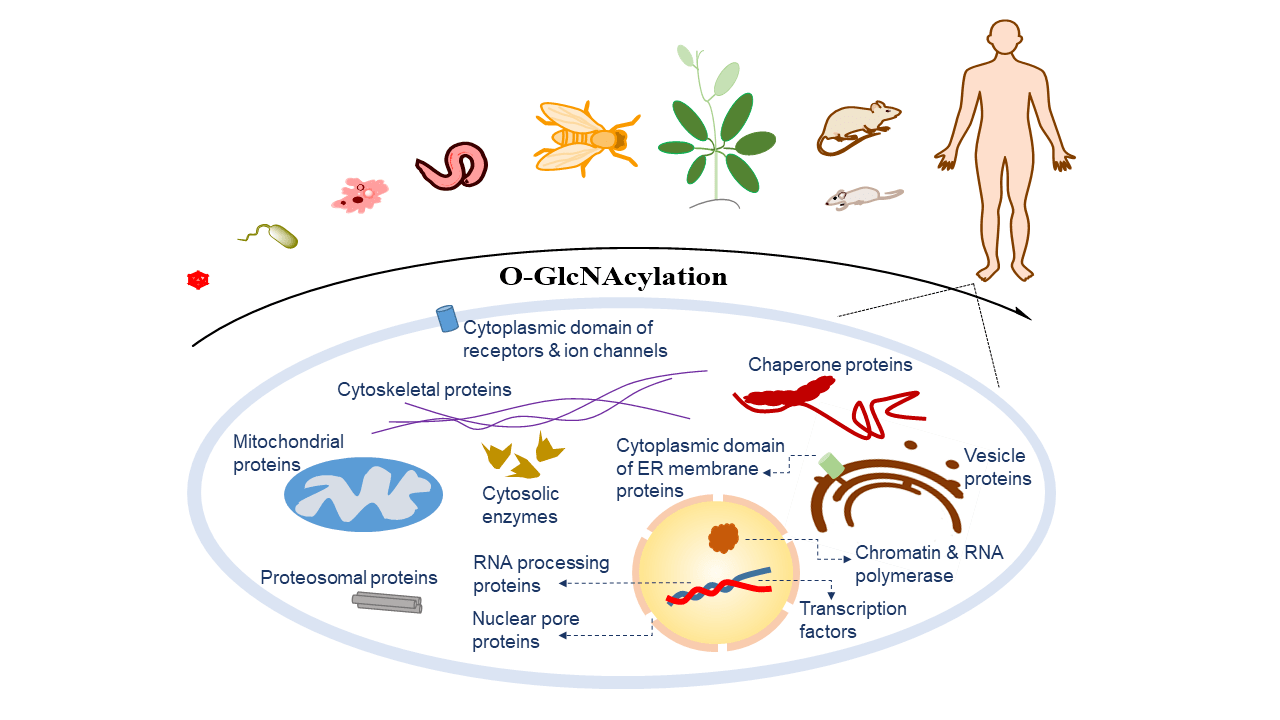
O-GlcNAcAtlas
Search and browse experimentally identified O-GlcNAc proteins and modification sites.
OGT-PIN
Explore curated interaction proteins of OGT and orthologues across experimental reports.

HexNAcQuest
Distinguish O-GlcNAc and O-GalNAc signals in glycoproteomics analysis.
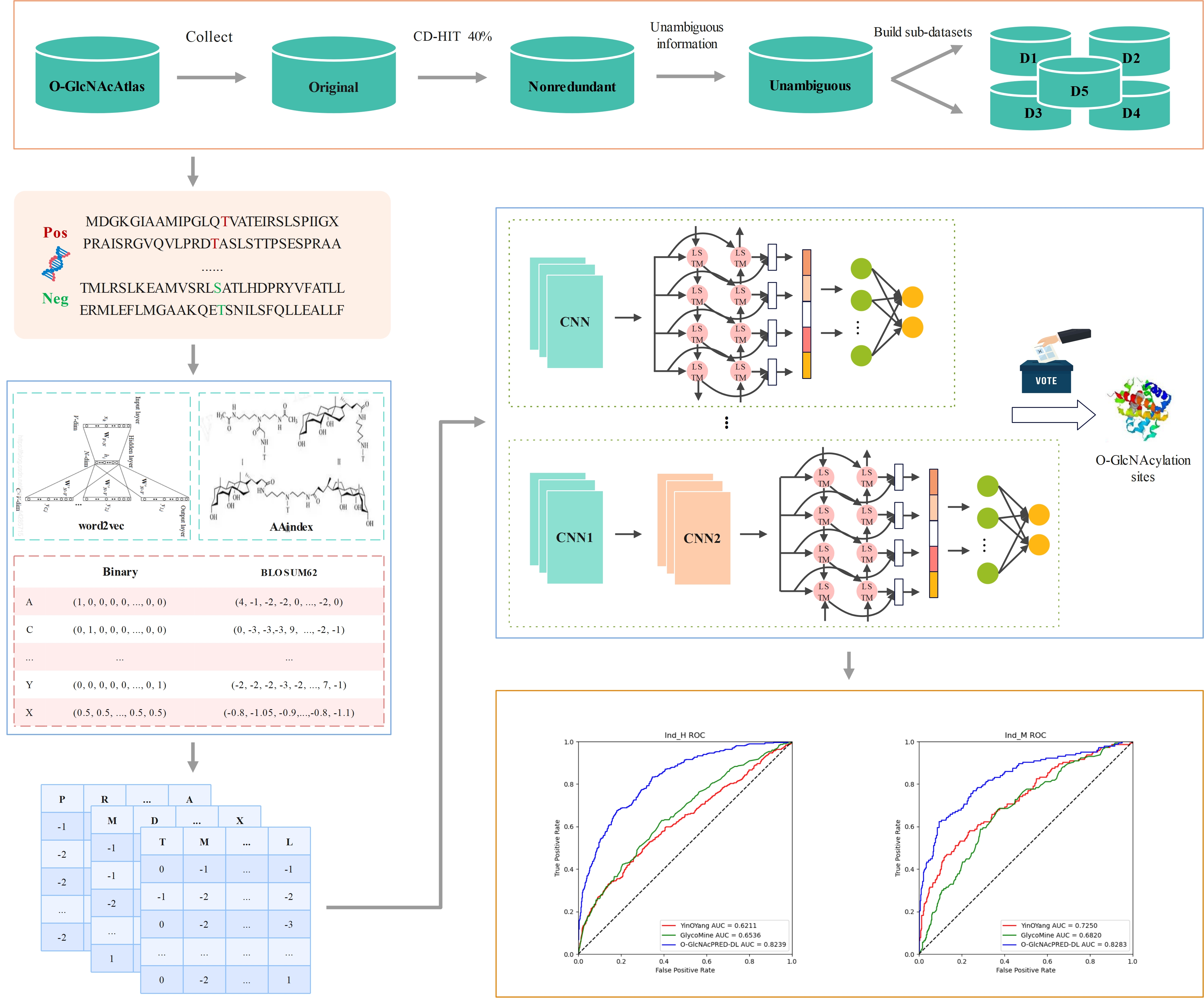
O-GlcNAcPRED-DL
Predict protein O-GlcNAcylation sites from FASTA input using ensemble deep learning.
Why It Matters
Dynamic glycosylation connected to signaling, disease, and therapeutic opportunity
O-linked beta-N-acetylglucosamine is a dynamic post-translational modification on serine, threonine, and tyrosine residues. It is regulated by OGT and OGA and has been implicated in transcription, translation, cell cycle control, metabolism, and signaling.
oglcnac.org brings together curated evidence and predictive tools so researchers can move quickly from a protein or sequence to sites, interactors, and candidate hypotheses.
Workflow
From query to biological hypothesis
- Search known O-GlcNAc proteins and modification sites.
- Inspect OGT interaction context and supporting identifiers.
- Submit FASTA sequences for site prediction when experimental evidence is absent.
- Download curated datasets for downstream analysis.

Start Here